Roles of clouds
Clouds play important roles in the Earth environment. They are;
1) Cloud amounts control albedo.
2) Clouds give influence to temperature through the process of greenhouse effect.
3) Clouds heat upper atmosphere by releasing latent heat
4) Clouds produce precipitation on lands and sustain life on lands.
How do these effects work on the cubic Earth? Understanding these processes will help us to understand atmospheric phenomena on the real Earth.
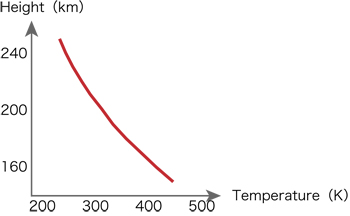
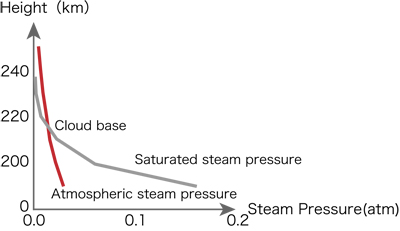
Can we see clouds in the sky of the cubic Earth? Fig.1 compares the water vapor pressure of the air on the cubic Earth with saturated water vapor pressure as a function of altitude. The water vapor decreases with altitude. The saturated water vapor pressure also decreases with altitude due to the temperature decrease. The water vapor is saturated at the height of 215km. There are cloud bottoms at this altitude. The temperature at this altitude is about 300K. Clouds cover in the form of a dome over squares. Clouds touch the ground at a point of 1,100 km apart from the center of the square. Probably, the ground outside of this area is covered by fogs. Fig. 2 shows the temperature near the cloud bottoms as a function of altitudes.
[Cloud amount]
Cloud amounts are defined by the ratio of cloud area to the whole sky. Cloud amounts “0” mean for no clouds, cloud cover “10” for being covered completely by clouds. In Japan, the cloud amounts are indicated by 12 levels, from 0 to 10, and 11 for “unclear”. When the condition of the sky is not observable due to mist etc., the cloud amounts will be the level 11 for “unclear”.
Cloud amounts “0” are clear day, “1~8” sunny day, “9, 10” cloudy.
[Latent heat]
Imagine putting water and ice into a beaker. The temperature of water in the beaker set to 0 ℃. Next, heat the beaker. Thermometer of the water in the beaker indicates 0 ℃ for a while. Thermal energies are given, because the beaker is heated. The temperature of the water should be increase due to increasing thermal energies.
Where have the added thermal energies gone? The added thermal energies have been used to melt ices. That is, thermal energies are needed to change from solid state to liquid one. It is called the melting heat.
Similarly, to change from liquid state to gas state, the thermal energies are needed. This is called vaporization heat. The latent heat means both melting heat and vaporization heat.
[Saturated vapor]
As you know, there are the dry air and wet one. The air will be dry in winter, on the other hand, it will be wet in rainy season. These are caused by amount of water vapor in the air.
How does air can contain the water vapor? The maximum amount of water vapor in the air depends on the air temperature. The maximum volume is called saturated vapor (the graph illustrated the maximum vapor [g] in 1m3 at each temperature).
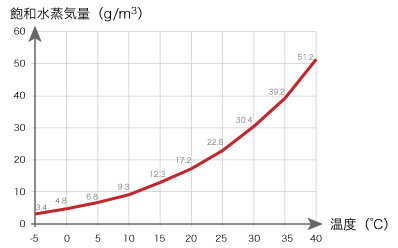
- Fig. 3
In saturated air at 20 ℃ of 1 m3, 17.2 g of water vapors are contained.
By the way, the temperature of the air decreases more and more, what is happened. Consider that the temperature of the air reaches 10 ℃. At its temperature, air of 1m3 can contain only 9.3 g of water vapor. Where has the water vapor of 7.9 g gone? The vapor has changed gas into liquid (water), appearing on the wall of glass.
In the warm room of winter, you can see the sweat on the windows. The sweats are changed from water vapor in the air whose temperature decreases toward the windows.



